Shop Products
Changing transfection plate format from a 6-well to a 96-well format
Moving a transfection reaction from a 96-well plate to a 6-well plate, or vice versa, and wanting to achieve identical results is not a trivial pursuit. It is well known that lipoplexes adhere to the plastic surfaces of cell culture wells. As one can conclude, this decreases the amount of complexed material freely available to transfect the cells. Many commercially available transfection reagent manufacturers provide a scaling factor between differing plate sizes; thus, setting up the researcher for failure. As seen in the table below, how do you chose a single ratio to determine transfection protocol scaling? For example, if up- or downscaling is based on either growth area, well volume or well diameter, one must consider whether the resulting working volume is enough such that the resultant miniscus in the well is appropriate as to not cause cells to adhere to the outer rim of the well. Not taken into consideration is the working volume depth, which varies between a 96-well and 6-well plate setup; thus, the exposed surface area on the side of the wells in which the lipoplexes adhere is altered. This is the reason why optimized parameters determined for a specific plate format cannot be applied as a set proportion to other vessel formats. Changing plate formats requires the conditions to be re-optimized for the new plate size.
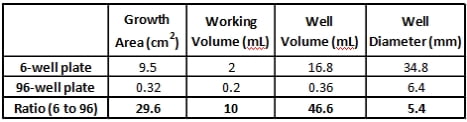
Table 1: Cell culture plate dimensions and working volumes. The ratio on bottom row was determined as a ratio of the 6-well value to the 96-well value. It is apparent that a single parameter cannot be used for transfection scaling purposes.




Sorry, comments are closed for this post.