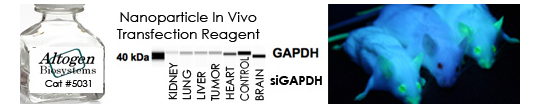
Shop Products
What is the best antibiotic to use for stable cell selection in mammalian cells?
There is a long list of antibiotics available to researchers when applying selective pressure in the creation of a stable cell line. The choices include zeocin, hygromycin, blasticidin, puromycin and geneticin (G418). Researchers use different antibiotics due to cost or availability in their lab. However, in reference to using antibiotics for the creation of a stable cell line, the antibiotic reference gene that is part of the plasmid construct must match the antibiotic used for selecting positive cells.
o Use 200 – 800 ug/ml of Neomycin (also called Geneticin or G418) for mammalian cells selection. While HeLa cell is efficient at 200 ug/ml of G418, other cells may require substantially higher concentration of geneticin (for example, optimal G418 concentration for A549 cell line selection is 800 ug/ml). Typical G418 cell line selection:
| Cell Line | CHO | DU145 | HepG2 | MCF-7 | PC-12 | SK-N-MC | SK-N-SH |
| G418 (µg/ml) | 900 | 200 | 700 | 800 | 500 | 900 | 1000 |
o Use fresh working stocks since most antibiotics quickly breakdown in solution. Store aliquots at -20°C and avoid freeze/thaw cycles
o Ampicillin quickly breaks down and plates should be used within 1 month for optimal efficiency
o Carbenicillin is very stable and can be used in place of Ampicillin in most applications
o Antibiotics are subject to degradation; do not add to media above 55°C and store stocks and agar plates wrapped in foil
o Some E. coli strains have antibiotic resistances; ensure your plasmid construct and E. coli strain are compatible
Researchers often use GFP expressing plasmid as a control in their transfection experiments. Plasmid DNA vector that encodes GFP (or usually eGFP) in its backbone also express ampicillin (Amp) or kanamycin (Kan) antibiotic resistance gene. However, it is important to know that both ampicillin (Amp) or kanamycin (Kan) are completely ineffective for mammalian cells selection (they can only be used for bacteria). Mammalian cells selection requires use of puromycin, geneticin (G418), zeocin, hygromycin, or blasticidin.