Shop Products
How do I increase transfection efficiency?
Transfection is considered as a major laboratory method to integrate protein, RNA and DNA molecules into tissues and cells. Delivery of plasmid DNA molecules containing gene inserts, messenger RNA and small interfering RNA molecules that regulate gene expression (i.e. microRNA, siRNA) into the primary cells and cancer cell lines have been extensively utilized by scientists. Transfection efficiency is an all-encompassing phrase that is used to superficially sum up the success of an experiment that contains many moving parts. Minimally, a successful transfection depends upon the optimization of various factors:
o cell density
o cell type
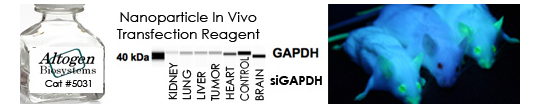
o transfection reagent
o transfection reagent volume
o nucleic acid concentration
o ratio of DNA to transfection reagent
o complex formation incubation time
o various media components (i.e. serum, antibiotics)
A lesser acknowledged consideration regarding transfection is the timing of the end-point analysis. Transient gene knockdown using siRNAs or miRNAs can exhibit desired results as early as 4 hours post-transfection, maximal effects at 24-48 hours and loss of transient effect around 96 hours. In contrast, the analysis of a plasmid DNA transfection may need to be altered to allow ample time for DNA uptake, expression and efficacy of the expressed target gene. The timing of these cellular events is often overlooked and too easily generalized as a “one-size fits all” theory. There are several pre-optimized transfection reagents commercially available from Altogen Biosystems (https://altogen.com/products-index/) for such cell lines as: MEF, A549, HepG2, Fibroblast, VERO, and many others. Also, in vivo transfection kits allow delivery of protein, DNA, and RNA molecules into animals (https://altogen.com/product/in-vivo-transfection-kits-peg-liposome-nanoparticle-polymer-lipid-tissue-targeted/).