Shop Products
Comparison of Transfection Reagents: siRNA and DNA
Introduction
The process of in vitro transfection involves the introduction of genetic material into cells and is generally used for mammalian cells through non-viral methods [1]. Various therapeutic cargo molecules can be used for intracellular delivery into cancer cell lines and primary cells, including plasmid DNA (pDNA), proteins, small molecules, messenger RNA (mRNA), and RNA such as short interfering RNA (siRNA) and microRNA (miRNA) [2]. Altogen Biosystems develops optimized transfection technologies for specific cell lines by applying expertise in combinatorial chemistry, molecular biology, and cell biology to the development of the transfection technology.
Transfection has been in use since the 1950s and remains a vital element in cell biology research with techniques ranging from physical techniques (such as electroporation) to chemically-mediated transfection using calcium phosphate, and even liposomal transfection technologies [3]. In liposomal transfection, the genetic material is contained in a liposome by mixing the material with a cationic lipid, and the liposome deposits its “cargo” into the target cell. Transfection reagents can be optimized to the target cell line and protocols for transfection can also be customized. Several advanced methodologies have emerged recently such as lipid and polymer-based carrier molecules. These compounds are capable of creating liposomes, which can fuse with the cellular membrane in order to deliver the bound RNA or DNA to the cell.
Transfection: Mechanism of Action
In vitro transfection is the transient or stable introduction of exogenous molecules and genetic material, DNA or RNA, into cultured mammalian cells. Although there is methodological diversity with transfection techniques, chemical transfection is the most prominent one that is used in current laboratory research. The transfection reagents used have cationic lipids that help in the delivery of DNA and siRNA into cells [4]; while cationic polymers were one of the earliest chemicals used, cationic lipids are now the most popular chemical in use today. The science behind chemical transfection remains the same, and it is based upon electrostatic interactions between cargo-carrying molecules and the lipid bilayer of the cell membrane. Essentially, negatively charged genetic material binds to positively charged reagents, allowing for the genetic material to pass through cell membranes, resulting in cellular uptake that occurs through endocytosis. Inside the cell, the transfection complex can serve a variety of purposes. If DNA is being transfected, then stable expression requires that it enters the nucleus, resulting in gene expression [5]. Factors that can affect the specific chemical choice depend upon the genetic material that one is transfecting, but ones such as pH, cell type, and ratio of the genetic material to reagent are critical to proper experimental setup.
Transfection Methods
Introducing DNA and RNA into cultured mammalian cells can require different transfection methods depending on the specific cell line and end goal. Such methods include liposome-mediated transfection, non-liposomal transfection agents (lipids and polymers), dendrimer-based transfection, electroporation, microinjection, virus-mediated gene delivery, RNA Interference (RNAi), siRNA Transfection, high throughput siRNA screens, and gene silencing (RNAi) [6, 7, 8].
Transfection Reagents
It is extremely important for chemical transfection experiments to retain cell viability; if the cells die as a result of toxic chemicals used for transfection, then the experiment is over. Transfection reagents are numerous precisely because certain cell types require milder conditions or special additives to prevent cytotoxicity. To this extent, Altogen Biosystems provides a comprehensive assortment of reagents designed for specific cell lines, designed specifically to improve transfection efficiency while preventing cell death.
Transfection reagents by themselves consist of specialized formulations of buffers optimized for specific cell types. In some cases, transfection reagents can include molecules that are designed to help desired compounds enter a cell. For example, a lipofection transfection reagent will include chains of liposomes that encapsulate a given nucleic acid sequence and allow it to be endocytosed by a cell. Another example is a transfection reagent made with nanoparticles that can bind to desired sequences and help them pass through the cell membrane.
An important distinction should be made between transfection reagents that bind to desired cargo and those that affect a cell membrane. An electroporation buffer, for example, is a transfection reagent that allows for a cell’s membrane to become permeable, hence allowing for foreign particles to enter the cell. However, electroporation buffers do not encapsulate nucleic acid sequences, and hence electroporation experiments must rely on the independent stability of transfected compounds.
In Vitro Transfection Studies
Altogen Biosystems provides different in vitro transfection reagents optimized for different cell lines such as A549 cells (lung carcinoma), DU145 cells (prostate carcinoma), MCF-7 cells (breast cancer), and HepG2 cells (hepatocellular carcinoma). Below is data compiled from several studies conducted with in vitro transfection reagents that are cell line-specific.
Transfection Reagent for A549 Cells (Lung Carcinoma Cells, CCL-185)
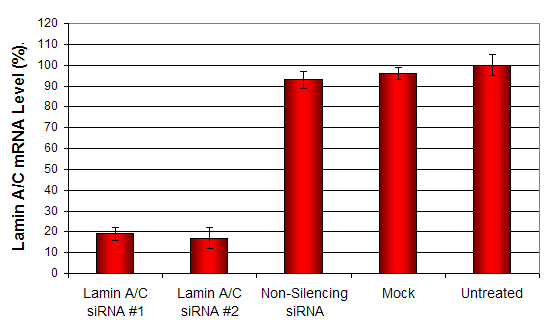
Figure 1. siRNAs targeting Lamin A/C mRNA or non-silencing control siRNA were transfected following the recommended protocol. At 48 hours post-transfection the A549 cells were analyzed by qRT-PCR for gene expression levels. 18S rRNA levels were used to normalize the Lamin A/C data. Values are normalized to untreated sample. Data are means ± SD (n=3).
- Two component formulation enhances lipid mediated transfection efficiency,
- Optimized easy-to-use transfection protocol provided for transfection of RNA and DNA
- High transfection efficacy in the presence of serum
- Works well for standard reverse transfection and high-throughput applications.
- Reagent exhibits at least 80% transfection efficiency of siRNA delivery. Transfection efficiency was determined by real-time RT-PCR.
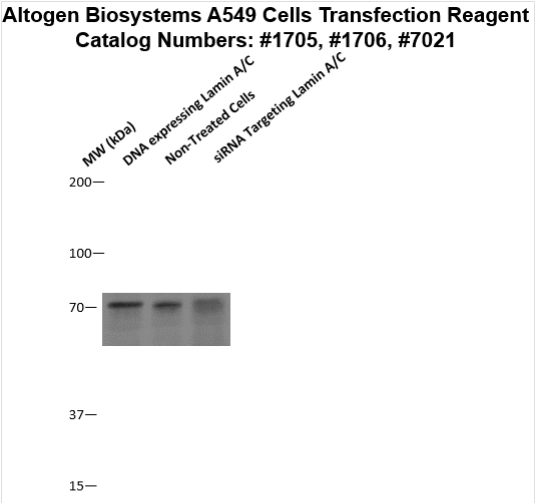
Figure 2. Protein expression of Lamin A/C in A549 cells. DNA plasmid expressing Lamin A/C or siRNA targeting Lamin A/C were transfected into A549 cells following Altogen Biosystems transfection protocol. At 72 hours post-transfection the cells were analyzed by Western Blot for protein expression levels (normalized by total protein, 10 µg of total protein loaded per each well). Untreated cells used as a negative control.
Transfection Reagent for DU145 Cells (Prostate Carcinoma Cells, HTB-81)
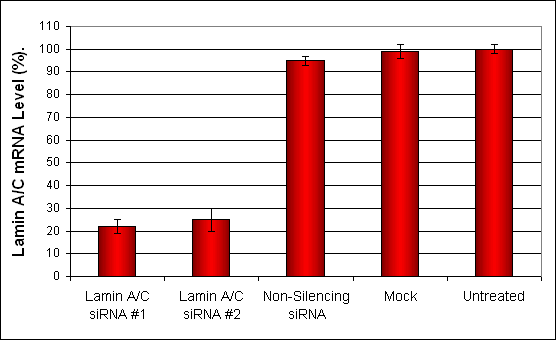
Figure 3. siRNAs targeting Lamin A/C mRNA or non-silencing control siRNA were transfected into DU-145 cells following the recommended protocol. At 48 hours post-transfection the cells were analyzed by qRT-PCR for Lamin A/C gene expression levels. 18S rRNA levels were used to normalize the Lamin A/C data. Values are normalized to untreated sample. Data are means ± SD (n=4).
- Proprietary cationic lipids formulation
- High transfection efficiency of small RNA (siRNA, shRNA, miRNA), mRNA, pDNA
- Produces consistent results, lot-to-lot, plate-to-plate, and well-to-well
- A proven reagent for establishing stable cell lines
- Optimized transfection protocols are adapted for use with both standard & reverse transfection methods.
- Effective and robust intracellular delivery
- Kit includes Transfection Enhancer reagent
- Work in the presence of serum
- Reagent exhibits at least 75% transfection efficiency of siRNA delivery. Transfection efficiency was determined by qRT-PCR.

Figure 4. Protein expression of Lamin A/C in DU145 cells. DNA plasmid expressing Lamin A/C or siRNA targeting Lamin A/C were transfected into DU145 cells following Altogen Biosystems transfection protocol. At 72 hours post-transfection the cells were analyzed by Western Blot for protein expression levels (normalized by total protein, 10 µg of total protein loaded per each well). Untreated cells used as a negative control.
Transfection Reagent for MCF-7 Cells (Breast Cancer Cells, HTB-22)
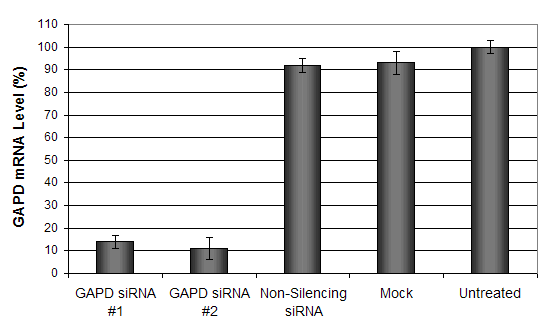
Figure 5. GAPD mRNA levels were quantified using real-time RT-PCR in the MCF-7 cells transfected with siRNAs targeting GAPD or non-silencing siRNA. Forty-eight hours post-transfection, the cells were harvested and analyzed by real-time RT-PCR for GAPDH mRNA expression levels. Data were normalized against the 18S rRNA signal. Control samples were either mock-transfected or untreated. Values are normalized to untreated sample. Data are means ± SD (n=3).
- Proprietary cationic lipids formulation
- High transfection efficiency of small RNA (siRNA, shRNA, miRNA), mRNA, pDNA
- Produces consistent results, lot-to-lot, plate-to-plate, and well-to-well
- Work in the presence of serum
- A proven reagent for establishing stable cell lines
- Reagent exhibits at least 85% transfection efficiency of siRNA delivery. Transfection efficiency was determined by qRT-PCR.

Figure 6. Protein expression of GAPDH in MCF-7 cells. DNA plasmid expressing GAPDH or siRNA targeting GAPDH were transfected into MCF-7 cells following Altogen Biosystems transfection protocol. At 72 hours post-transfection the cells were analyzed by Western Blot for protein expression levels (normalized by total protein, 10 µg of total protein loaded per each well). Untreated cells used as a negative control.
Transfection Reagent for HepG2 Cells (Hepatocellular Carcinoma Cells, HB-8065)
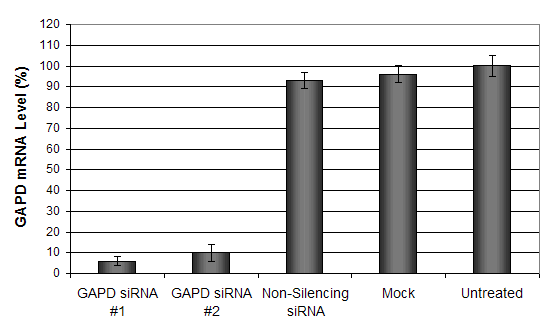
Figure 7. GAPD mRNA levels were quantified using real-time RT-PCR in the HepG2 cells transfected with siRNAs targeting GAPD or non-silencing siRNA. Forty-eight hours post-transfection, the cells were harvested and analyzed by real-time RT-PCR for GAPDH mRNA expression levels. Data were normalized against the 18S rRNA signal. Control samples were either mock-transfected or untreated. Values are normalized to untreated sample. Data are means ± SD (n=3).
- High transfection efficiency of small RNA (siRNA, shRNA, miRNA), mRNA, pDNA
- Effective and robust intracellular delivery
- Produces consistent results, lot-to-lot, plate-to-plate, and well-to-well
- Work in the presence of serum
- A proven reagent for establishing stable cell lines
- Reagent exhibits at least 90% transfection efficiency of siRNA delivery. Transfection efficiency was determined by qRT-PCR.

Figure 8. Protein expression of GAPDH in HepG2 cells. DNA plasmid expressing GAPDH or siRNA targeting GAPDH were transfected into HepG2 cells following Altogen Biosystems transfection protocol. At 72 hours post-transfection the cells were analyzed by Western Blot for protein expression levels (normalized by total protein, 10 µg of total protein loaded per each well). Untreated cells used as a negative control.
Conclusion:
Transfection is a well-tested and crucial biotechnology that allows researchers to study gene products and gene functioning directly in cells. The different transfection methods allow the possibility of delivering genetic material in a very specific manner, thus making this an extremely versatile tool in preclinical biology research. Choosing the optimum method involves implementing the best transfection reagent, as well as picking the best experimental design.
Altogen Biosystems offers pre-optimized and cell line-specific in vitro transfection reagents and kits. Learn more about these transfection reagents and kits on their website and expedite your preclinical biology research study today.
www.altogen.com/products-index/ (Altogen Biosystems offers 100+ of cell-line specific in vitro transfection reagents)
References:
- Hannon, G.J. and J.J. Rossi, Unlocking the potential of the human genome with RNA interference. Nature, 2004. 431(7006): p. 371-8.
- Schlake, T., et al., Developing mRNA-vaccine technologies. RNA Biol, 2012. 9(11): p. 1319-30.
- Kittler, J.T., J.G. Hanley, and J.T.R. Isaac, Transfecting and Transducing Neurons with Synthetic Nucleic Acids and Biologically Active Macromolecules
- Yin, H., et al., Non-viral vectors for gene-based therapy. Nat Rev Genet, 2014. 15(8): p. 541-55.
- Kaestner, L., A. Scholz, and P. Lipp, Conceptual and technical aspects of transfection and gene delivery. Bioorganic & medicinal chemistry letters, 2015. 25(6): p. 1171-6.
- Chang, F.H., et al., Surfection: a new platform for transfected cell arrays. Nucleic acids research, 2004. 32(3): p. e33.
- Wang, K., et al., Non-viral Delivery Systems for the Application in p53 Cancer Gene Therapy. Current medicinal chemistry, 2015. 22(35): p. 4118-36.
- Rettig, G.R. and M.A. Behlke, Progress toward in vivo use of siRNAs-II. Molecular therapy : the journal of the American Society of Gene Therapy, 2012. 20(3): p. 483-512.