Shop Products
RNAi Therapeutics
RNAi Therapeutics
RNAi Therapeutics refers to the use of RNAi technology to treat genetic diseases and certain disorders. By changing the protein expression in cells, some genetically-linked diseases can at least be temporarily cured; the proteins expressed by cells can have a significant impact on overall tissue functionality.
Summary of RNAi Functionality
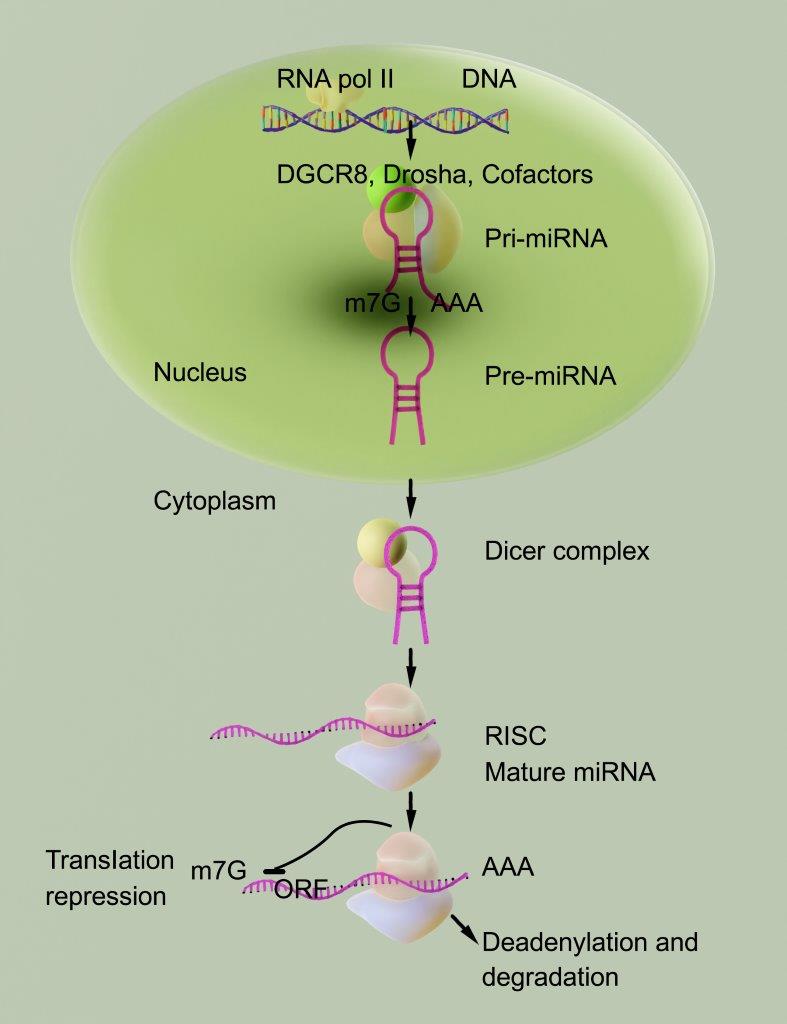
RNAi (RNA interference) is a process or mechanism that is found in all living cells and is responsible for inhibiting the “expression” or further development or behaviors of specific genes. This inhibition, in turn, affects the production, or lack thereof, of certain proteins.
Also known as therapeutic gene silencing, RNAi research has come far in the past decade. Since its discovery in 2001, greater understanding regarding the processes and potential of RNAi technologies in the treatment of diseases has continued. RNAi technologies are now focusing on state-of-the-art and innovative treatments and therapies in the treatment of a number of disease processes including cancers, infectious diseases, and obesity. In the medical and pharmaceutical industry, the focus on the development of effective drugs that specifically target a particular gene may provide huge strides in the treatment, and even potential reversal, of certain disease processes. In addition, the ability to specifically target genes that cause cellular damage or destruction leading to disease processes enables pharmaceuticals and therapeutics to specifically target genes that have previously been unaffected by a number of traditional drug therapies.
Applicability to Medicinal Fields
RNAi technologies can be utilized for the treatment of diseases by the effective silencing of gene expression and production of genes that cause disease (such as oncogenes). The degree of specificity in targeting such genes is another advantage of RNAi therapeutic modalities, as is the ability to utilize numerous RNA sequences in a specific or singular drug product in targeting and delivering drugs or medicines to cell-specific tissues or organisms. The ultimate goal for researchers is to find ways to damage, slow down, or kill defective and cancerous cellular proliferation without damaging or otherwise affecting surrounding tissues. Numerous patents in the process of further developing siRNA drugs and pharmaceuticals or small molecules and antibodies have been filed by biotechnology and pharmaceutical companies globally. Targeting RNAi-based therapeutics in the treatment of infectious diseases such as hepatitis, metabolic conditions such as obesity, and cancerous tumor growth is the focus of many companies who are sponsoring novel RNAi therapeutics through pre-clinical research efforts and clinical trials.
Barriers to Clinical Application
Research scientists use RNAi biological phenomena to study particular genes and their functions. The main hurdle in using RNAi for therapeutic gene silencing is the effective intracellular delivery of siRNA into the cell or tissue. The capability of siRNAs to mediate post-transcriptional gene silencing in mammalian cells and tissues, as well as its successful use to prevent expression of target mRNA has recently led to the development of a new methodology for novel drug discovery.
Several pharmaceutical and biotechnology companies are currently investigating the possible use of synthetic siRNA for inducing RNAi in vivo, its use in animal models, and in RNAi-based therapeutics. Small RNA molecules, such as siRNA, shRNA, and microRNA have been regarded as potential therapeutic agents to target multiple mis-regulated cellular processes therefore it is theoretically possible that RNAi can be utilized to treat any disease associated with over expression of specific genes. In fact, there are many reports that address the potential therapeutic application of RNAi to specifically target genes involved in multiple diseases including various forms of cancer, Alzheimer’s, and a number of inflammatory and virally-associated diseases.
Other applications of RNAi include the targeted inhibition of protein expression where it is necessary. In diseases such as achondroplasia, a functioning protein is simply over-produced. As such, the protein must not be completely eliminated; rather, the expression of the protein must be limited in a stable manner. RNAi, a technology capable of reducing protein expression for long periods of time, thus has the potential to cure diseases associated with gene over-expression.
Before RNAi becomes a common commercial therapeutic technology, a number of major difficulties associated with inefficient delivery of functional RNA molecules into cells and the reduced biostability of unmodified RNA must be overcome. Altogen Biosystem’s research is focused on the development of efficient in vivo reagents and RNAi delivery technologies. Efficient and organ-specific delivery of synthetic oligonucleotide molecules is currently a key limiting step to enable siRNA- and microRNA-based therapeutic approaches.
However, a number of major difficulties associated with inefficient delivery of functional RNA moleculesinto cells and the reduced biostability of unmodified RNA must be overcome. Therefore, our research is focused on the development of efficient in vivo reagents and RNAi delivery technologies. Efficient and organ-specific delivery of synthetic oligonucleotide molecules is currently a key limiting step to enable siRNA- and microRNA-based therapeutic approaches.
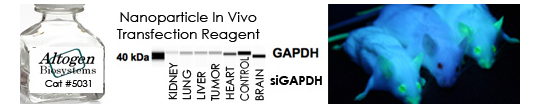
In Vivo Transfection Kits from Altogen Biosystems
RNAi has been used for in vivo target validation studies using animal models. The major challenge in performing RNAi studies in vivo is the effective, directed delivery of functional siRNA molecules into specific tissues. Altogen® in vivo transfection reagents could be conjugated with siRNA and administered systemically via intravenous (i.v) tail vein injection in order to provide directed gene silencing in specific tissues, including liver, pancreas, kidney, and tumors. Selective knockdown could be seen as early as 24 hours after injection.
Featured transfection products from Altogen Biosystems:
LNCaP Cell Transfection Kit || In Vivo Nanoparticle Transfection Kit || In Vivo PEG-Liposome Transfection Kit
Altogen Biosystems is a life sciences company focused on development, marketing and manufacture of cell type specific transfection products. Efficient delivery of DNA, RNA, and siRNA enabled by advanced formulation of reagents and peculiar design of protocols. Altogen Biosystems offers a complete transfection system for over 100 cell lines, as well as hard-to-transfect and primary cells. All reagents are functionally tested to be highly reproducible, serum compatible, induce low toxicity, and can be used for co-transfection experiments, and high throughput applications.
What is RNAi Therapeutics
RNA interference (RNAi) therapeutics is a type of gene therapy that uses small RNA molecules to target and silence specific genes involved in disease. RNAi is a natural mechanism by which cells regulate gene expression, and has been harnessed as a powerful tool for gene silencing.
RNAi therapeutics work by using small RNA molecules, such as short interfering RNA (siRNA) or microRNA (miRNA), that can specifically target and bind to messenger RNA (mRNA) molecules that encode for disease-causing genes. Once bound, the RNAi molecules can induce the degradation of the target mRNA molecules or inhibit their translation into proteins, leading to the suppression of the target gene expression and potentially treating the disease.
RNAi therapeutics have shown promise in the treatment of various diseases, including genetic disorders, viral infections, and cancer. They offer several advantages over traditional gene therapy approaches, such as their specificity and versatility, as they can target a wide range of genes and be delivered to various tissues and organs.
However, RNAi therapeutics also face several challenges, such as their delivery to specific cells and tissues, stability and off-target effects. Advances in technology and delivery systems, such as nanoparticles or viral vectors, are being developed to overcome these challenges and improve the efficiency and safety of RNAi therapeutics.
Overall, RNAi therapeutics hold great potential as a powerful tool for gene therapy and the treatment of various diseases, and ongoing research and development are expected to lead to new RNAi-based treatments in the future.
RNAi Services by Altogen Labs CRO:
Altogen Labs provides all types of RNAi laboratory services, as well as other specialized biotechnology and pharmaceutical services, including generation of stable cell lines, RNA interference (RNAi) services, pharmacology and toxicology testing: IC-50, LD-50, qPCR, automated Western Blot analysis, assay development, siRNA library screening and transfection services, liposome encapsulation, cell banking and cryopreservation services.