Shop Products
What transfection controls do I need to include in my experiment?
Scientists understand the importance of controls in experiment. There are at least three transfection controls that should be included on every transfection plate: a positive control, negative control and non-treated control.
Positive Controls
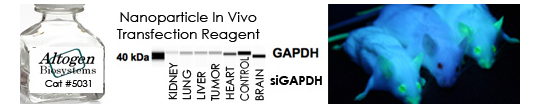
Transfecting a positive control ensures that the system being utilized is working and the delivery conditions are optimal. The results of the positive control aids the researcher in understanding any intra- and inter-experimental differences. The ideal positive control consists of these characteristics:
- Considered a housekeeping gene: This ensures the target gene is abundantly expressed in all cell types and at a constant level not induced by cell cycle
- Abundant expression: Enables accurate and easy quantitation of mRNA or protein levels
- Non-lethal: Silencing of the positive control expression should not affect the cells phenotype or viability
- Common targets: Glyceraldehyde-3-phosphate dehydrogenase (GAPDH), Cyclophilin B (PPIB), and Lamin (LMNA)
Negative Controls
Negative controls are designed to have no known computationally derived target in the cells being transfected and are commonly called a scrambled control. Ideal negative control design mimics the test article in length, G/C content and modifications. They are used for differentiating sequence-specific silencing from non-specific effects. All negative control samples are analyzed similarly to the samples treated with the test article. However, the ideal negative control exhibits these characteristics:
- Gene targets: Designed as an alien sequence (no known target) as to not affect mRNA or protein expression levels
- Non-lethal: Should not affect cell viability or global gene expression due to non-target design
Additional controls include a mock transfection (i.e. transfection reagent only) and non-treated control. The mock and scrambled control helps in understanding experimental toxicity when compared to the non-treated sample.




Sorry, comments are closed for this post.