Shop Products
Transfection optimization
The most vital aspect of a transfection is ensuring all conditions are optimized, including transfection reagent volume, oligo concentration, cell viability and passage number, lack of negative control activity, activity of positive control and calibrated incubator temperature, humidity and percentage of CO2. Even if all these parameters are correctly addressed, transfection efficiency must be determined using a quantitative assay. Optimized conditions are determined as the parameters that exhibit the highest resulting efficacy and the lowest toxicity relative to the non-treated cells. Here are some commonly utilized assays to assess transfection efficiency during transfection optimization:
- Reporter assay: A plasmid reporter encoding a fluorescent protein (i.e. GFP) is transfected and the resultant fluorescence is measured on a plate reader or cell imager
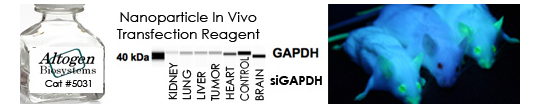
- Altered gene expression: An siRNA targeting a known, ubiquitously expressed gene (i.e. GAPDH, CyclophilinA, β-Actin) is transfected and resultant cell lysates are analyzed for changes in the target gene protein (e.g. western blot, ELISA) or mRNA levels (e.g. qRT-PCR)
- Co-transfection of an siRNA and plasmid: A plasmid containing a reporter gene (i.e. GFP) is co-transfected with an siRNA targeting the reporter gene; target gene knockdown fluorescence is measured relative to the negative control siRNA control fluorescence




Sorry, comments are closed for this post.