Shop Products
Cell Transfection
Cell transfection is the deliberate artificial introduction of nucleic acid (DNA or RNA) into living cells. Typically, this process is utilized to study gene function and protein expression. Sometimes confused with the term “transformation”, transfection is nearly always used in eukaryotic cells, while transformation is commonly referred to in bacterial work.
Transient and Stable Transfection
It is important to understand both transient and stable transfection techniques, and considering their role in the end product. Transient transfection is typically used for short term observations, while stable transfection is used for longer term gene studies. Furthermore, stable and transient transfections differ significantly in terms of the transfection process; in transient transfections, it is not necessary for transfected nucleic acids to enter the nucleus, while in stable transfections DNA must be incorporated into a cell’s genome.
History
The transfection method began as a research technique in the 1970’s when Vaheri and Pagano used DEAE-dextran, and Graham and Van der Eb used calcium phosphate for cell transfection experiments. These chemical-based methods were revolutionary at the time as they did not have to rely on viral vectors to introduce genetic material into cells. Once PCR was discovered, this accelerated the development of applications to introduce different types of genetic material into cells.
The modern era of cell transfection deals with introducing cargo such as plasmid DNA, mRNA, proteins, and small RNA (siRNA, microRNA, piRNA) into cultured cells in vitro and targeted tissue delivery in vivo. Experiments are designed and performed by testing the effect of these cargo molecules in cells. However, delivery efficiency depends on the specific transfection reagent used. Transfection reagents are specifically designed, developed, and optimized for each type of cancer cell, difficult-to-transfect cell lines, sensitive cells (such as primary and stem cells, as well as hormone-dependent neuronal cells, primary keratinocytes and aortic endothelial cells), adherent and suspension culture (such as primary T-cells).
Transfection Experiment
The goal in any transfection experiment is high transfection efficiency, meaning a good expression of the construct with minimal cytotoxicity. Achieving minimal stress to cells and reproducibility across experiments is influenced by the reagent used, and optimization of the transfection protocol. When transfecting DNA or RNA, the quality and purity of nucleic acid also has an influence on transfection efficiency. Also, a low passage number of cell line allow cells to be more amenable to uptake the transfection complex with cargo nucleic acid. There is no single method which can be reproduced across cell types and experiments. Each individual transfection must be optimized. However, cell line specific transfection kits with optimized transfection protocols are available from Altogen Biosystems.
Primary cell cultures are used in biological and gene therapy studies and serve as important model systems that may more accurately represent the biology of normal cells. Many cultured cell lines, as well as the majority of primary cell cultures, are able to be transfected with exogenous nucleic acids when appropriate transfection approaches are employed. Since the majority of transfection methods causes significant toxicity in primary cell cultures, optimizing this procedure (specifically the protocol and reagents to be utilized) is essential for developing effective transfection strategies for a given cell type. Altogen kits for primary cells and sensitive cell lines have been designed to have significantly lower cytotoxicity than other alternatives.
Transfection Methods
Transfection generally pertains to the chemical means of delivering nucleic acids into cells. Other techniques also exist and can accomplish similar goals. Viral-mediated transduction, for example, can deliver nucleic acids via modified viruses. The technique has shown significant effectiveness, and was the foundation of the first in vivo gene therapy approved by the FDA. However, transduction carries its side effects; immune responses from a patient can significantly alter the results of an introduced sequence of nucleic acids, making the effects differ from those observed in vitro.
Other transfection techniques can be based on other properties of cells and means of accessing the interior of a cell. Ballistic transfection techniques shoot DNA that is bound to gold particles at cells, with the aim of having the particles deliver the DNA into the nucleus. Other techniques such as electroporation make the cell membrane rip apart at places, allowing the introduction of foreign materials into cells. Chemical transfection however, is a technique that is generally less-invasive than alternatives, and hence has a lower potential for negative side effects.
In Vivo Transfection Reagents from Altogen Biosystems
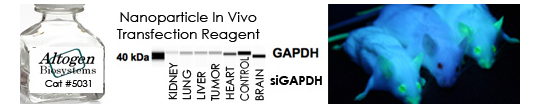
RNAi has been used for in vivo target validation studies using animal models. The major challenge in performing RNAi studies in vivo is the effective, directed delivery of functional small RNA molecules into specific tissues. Altogen® In Vivo Transfection Reagents could be conjugated with siRNA (or microRNA) and administered intratumorally (i.t) or systemically via intravenous (i.v) tail vein injection in order to provide directed gene silencing in specific tissues, including liver, pancreas, kidney, and tumors. Selective knockdown could be seen as early as 24 hours after injection.
What is Cell Transfection?
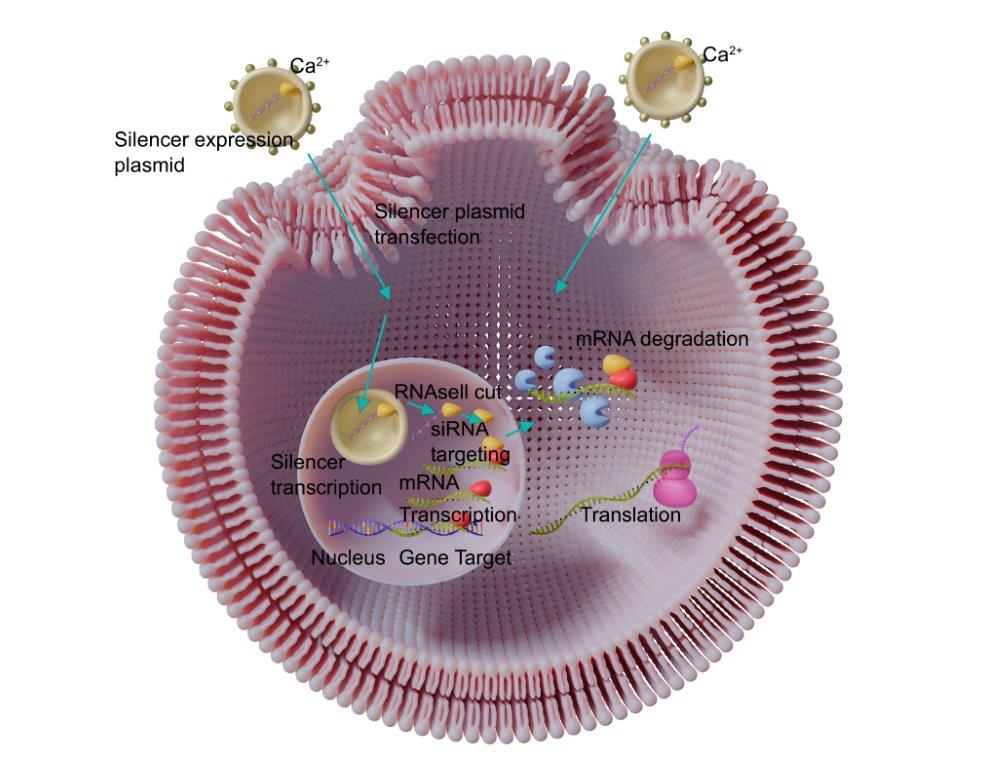
Cell transfection is a laboratory technique used to deliver nucleic acids, such as DNA, RNA, or siRNA, into cells to modify their gene expression or to study gene function. The technique involves introducing the nucleic acids into cells using a transfection reagent, which facilitates their entry into the cells.
Cell transfection typically involves the following steps:
- Selection of transfection reagent: Researchers select a transfection reagent that is compatible with the type of cells being transfected and the type of nucleic acid being delivered.
- Preparation of nucleic acids: The nucleic acids are prepared, either by isolating them from natural sources or synthesizing them in the laboratory.
- Formation of transfection complex: The nucleic acids are mixed with the transfection reagent to form a complex that can enter cells.
- Transfection of cells: The transfection complex is added to the cells, and the cells are incubated to allow the nucleic acids to enter the cells.
- Analysis of transfection efficiency: The efficiency of transfection is evaluated by measuring the level of gene expression of the delivered nucleic acid using techniques such as PCR, western blotting, or fluorescence microscopy.
Cell transfection is a valuable tool for studying gene function and for developing new therapies for genetic diseases or cancers. However, the efficiency and specificity of transfection can be affected by various factors, including the choice of transfection reagent, the type of cells being transfected, and the experimental conditions. Therefore, careful optimization of transfection conditions and appropriate controls are important to ensure the reliability and reproducibility of transfection experiments.
Altogen Research Services
Altogen Custom Services provides cell transfection services and other specialized biotechnology and pharmaceutical services, including over 60 validated xenograft models, development of stable cell lines, RNA Interference (RNAi) services, assay development, ELISA and Western Blot services, siRNA library screening and transfection services. Generation of stably-expressing cell lines can be very expensive and time-consuming. Altogen Labs offers generation of stable cell line service completed in just 28 days (see service details).