Shop Products
Quantitate in vitro anti-proliferation experiments using a metabolic assay (Alamarblue, MTT) vs measuring protein abundance (Sulforhodamine B)
The need to measure cell proliferation effects is vast, including testing the effects of growth factors, novel pharmacological agents, cytotoxicity assessment or investigating cell activation. Cell proliferation assays utilize a stain or substrate to make a correlation between the readout and the number of remaining cells post-treatment. However, the means of assessing cell number varies greatly.
Metabolic Assays
Metabolic assays rely on proliferating cells to metabolize a substrate, thus, producing a color change of the cell media. The resultant color change or fluorescence measurement is used to make a correlation to a change in cell number. The MTT assay is by far the most popular, but being insoluble in culture media, it is an end-point assay. On the other hand, the Alamarblue assay (all rights belong to Thermofisher) is soluble in media, is non-toxic and can be applied to cells multiple times throughout the experiment for multiple measurements on the same wells.
Protein Abundance Assay
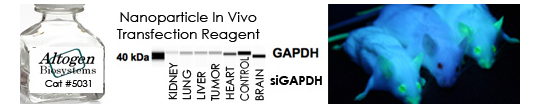
As the name implies, the abundance of all proteins is quantified in this assay. The protein dye sulforhodamine B, referred to as SRB, binds electrostatically to proteins. The dye interaction is pH dependent on the fixed amino acid residues. The protocol consists of four main steps: 1) preparation of cells, 2) treatment of cells with test article, 3) fixation and SRB staining, and 4) measure absorbance on a plate reader. Results of the SRB assay correlate with cell number and assay sensitivity is comparable with commercially available fluorescence assays. The SRB assay can be utilized in evaluating the effects of gene expression modulation (i.e. gene knockdown, gene upregulation) in 96-well or 384-well format.




Sorry, comments are closed for this post.